News
Those linked to stem cell board received more than $2.1 billion
 A medical researcher examines cancer stem cells. (Photo: luchschenF, via Shutterstock)
A medical researcher examines cancer stem cells. (Photo: luchschenF, via Shutterstock)Over the last 15 years, California’s stem cell agency has spent $2.7 billion on research ranging from arthritis and blindness to cancer and incontinence. The vast majority of the money has gone to enterprises that have ties to members of the agency’s governing board.
All of which is legal. All of which is not likely to change.
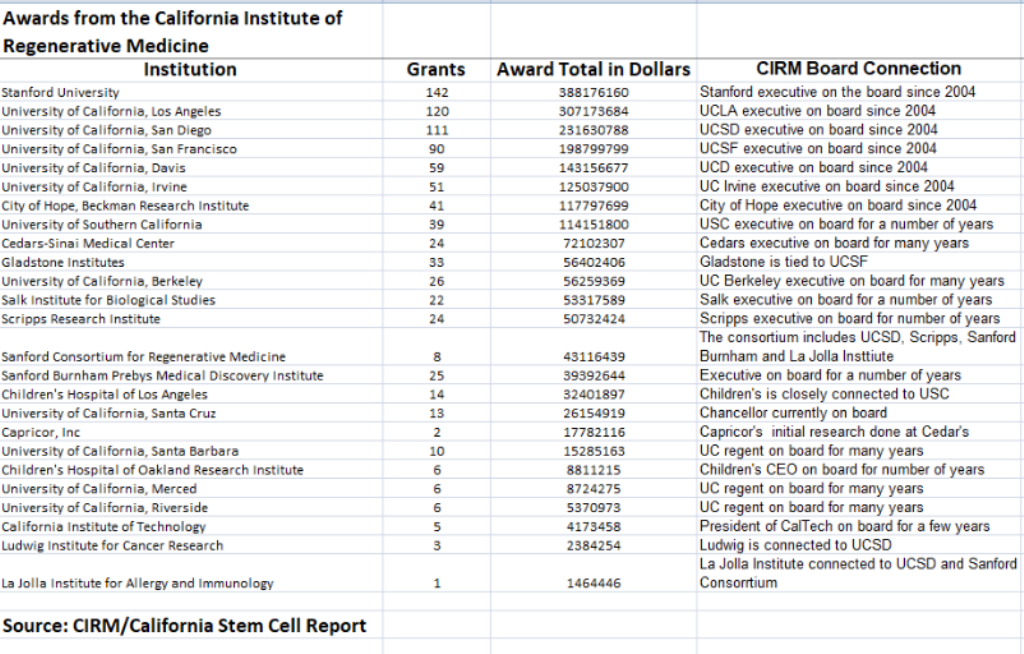
Eight out of every ten dollars that agency has handed out have been collected by 25 institutions such as Stanford University, multiple campuses of the University of California and scientific research organizations. Their combined total exceeds $2.1 billion.
All 25 have links — directly or indirectly — to past or present members of the board of the agency, according to an analysis by the California Stem Cell Report, which has covered the agency since 2005.
“They (the agency’s directors) make proposals to themselves, essentially, regarding what should be funded. They cannot exert independent oversight,” says Harold Shapiro, who led a 2012 study of the agency by the prestigious Institute of Medicine (IOM), which is now called the National Academy of Medicine. The study recommended a major restructuring of the agency’s board to help deal with the problem.
The longstanding, conflict-of-interest issues are not addressed in Proposition 14 on the Nov. 3 ballot. The measure would give the agency, officially known as the California Institute for Regenerative Medicine (CIRM), $5.5 billion more and expand its scope of activities and research. The ballot measure is likely to increase the problems by increasing the size of the agency’s governing board from 29 to 35.
Another ballot initiative, Proposition 71, created California’s stem cell program in 2004. Ever since, conflict of interest questions have dogged CIRM. Indeed, critics of the agency can today point to the top five recipients of CIRM largess as examples of conflict problems. Stanford University ranks as the No. 1 recipient with $388 million. UCLA is No. 2 with $307 million. It is followed by UC San Diego, $232 million; UC San Francisco, $199 million, and UC Davis, $143 million.
All have had a representative on the CIRM board since the inception of the program.
 Editor’s note CIRM’s totals may change slightly as the result of the agency’s internal accounting procedures.
Editor’s note CIRM’s totals may change slightly as the result of the agency’s internal accounting procedures.
IOM and public confidence in CIRM
The IOM study, with its criticism of conflicts, was commissioned by CIRM at a cost of $700,000. Directors expected that it would provide a “gold standard” evaluation of the agency that would support a ballot measure for additional funding. The study’s scope went well beyond conflicts of interest. In fact, it said it did not search for evidence of specific conflicts because the task was not part of the agreement with CIRM. The IOM did say that “studies from psychology and behavioral economics show that conflict of interest leads to unconscious and unintentional ‘self-serving bias’ and to a ‘bias blind spot’ that prevents recognition of one’s own bias.” While all of the study’s findings were consequential, the matter of conflicts attracted the most public attention.
“Ties to stem cell board lucrative,” said a headline in the Orange County Register shortly after the IOM report was released.
“The agency has used more than half of its funding and one day will almost certainly want to ask taxpayers for more. It should remember that voters will look for evidence of public accountability as well as respected research,” said the Los Angeles Times in an editorial in December 2012.
The IOM report itself said, “Far too many board members represent organizations that receive CIRM funding or benefit from that funding. These competing personal and professional interests compromise the perceived independence of the ICOC (the CIRM governing board), introduce potential bias into the board’s decision making, and threaten to undermine confidence in the board.”
The IOM said the composition of the board makes it neither “independent” nor capable of “oversight,” although the board is legally dubbed the Citizens Independent Oversight Committee (ICOC).
Placing deans of medical schools and patient advocates on the board who are linked to specific diseases “raises questions about whether decisions delegated to the board—particularly decisions about the allocation of funds—will be made in the best interests of the public or will be unduly influenced by the special interests of board members and the institutions they represent. Such conflicts, real or perceived, are inevitable….”
The situation involves more than legalisms. “Properly understood,” the IOM said, “conflict of interest is not misconduct, but bias that skews the judgment of a board member in favor of interests that may be different from or narrower than the broader interests of the institution.”
The IOM study additionally surveyed board members about conflicts of interest and reported, “While a majority of respondents stated that personal interests did not play a role in their work on the ICOC, some responses were more equivocal. One respondent replied that it was ‘hard to tell’ given that so many decisions take place off camera in secret meetings,’ while another acknowledged that ICOC members are human, and, of course, their decisions are influenced by personal beliefs and interests.”
The ‘inherent’ conflicts
The conflicts were built in by Proposition 71, which dictated the composition of CIRM’s 29-member board. CIRM’s general counsel, James Harrison, once described the situation as “inherent conflicts of interest.”
Under Proposition 71, representatives from virtually all the California institutions that stood to benefit were given seats at the table where spending plans are approved and awards handed out. Directors are not allowed to vote on specific awards to their institution. But they control the direction of the agency and what CIRM calls “concept” plans, including specific elements and budgets for the award rounds. Some of those rounds run into hundreds of millions of dollars.
One of the “concept” plans created a $47 million program to help California institutions recruit star scientists to the Golden State. Another plan created the $50 million Alpha Clinic Network at five academic centers all connected to board members.
Following the IOM report, the CIRM board did remove most institutional directors from meetings where awards are ratified. Jonathan Thomas, chair of the board, declared then that financial conflict issues were “put to bed once and for all,” a position that the agency holds today. In May 2019, Thomas told directors that several “authoritative entities” have studied CIRM and produced written reports that dealt with conflict matters.
Thomas said, “Each had in it sort of quite vehement language about the conflict of interest issue, which has always been just perceived…..With respect to any given funding award, there’s never been an actual conflict.”
During the 2019 meeting, the board did not discuss issues involving board action on “concept” plans. They continue today to modify and approve “concept” plans.
Beyond the CIRM board
Conflicts of interest at CIRM go beyond the 29-member board. In 2014, the agency was shocked by a case involving a former president of the agency, Alan Trounson, and StemCells, Inc., a company that was awarded $40 million while he was serving as the top executive at CIRM. (The company later declined one of the awards.) Only seven days after his final day at CIRM, Trounson was named to the board of directors of StemCells, Inc.
He served on the company’s board for about two years and received $443,500 in total compensation, including stock options, according to StemCells, Inc., documents filed with the Securities and Exchange Commission.
Following the announcement of the Trounson appointment, CIRM looked into some of Trounson’s work at CIRM. In July of 2014, the agency said that its “severely” limited investigation found no evidence that its former president attempted to influence action on behalf of StemCells, Inc., during the previous month. The state’s political ethics agency, the Fair Political Practices Commission, said in a Feb. 6, 2015, letter to Trounson that there was “insufficient evidence to demonstrate” a legal violation.
Even before the agency was created, critics warned of conflict-of-interest problems. Writing in an opinion piece in October 2004 in the San Francisco Chronicle, David Winickoff, then a professor at UC Berkeley, said, “Contrary to what its name suggests, the ICOC is neither ‘independent’ of interest-group politics nor does it include any ‘citizen’ members. Hard- driving university scientists, disease group advocates and private industry executives who will make up the ICOC all have vested interests in how the money is to be used.”
A sampling of conflicts
The California Stem Cell Report, which calculated the percentage of awards linked to institutional directors, has chronicled the conflicts issues at CIRM over the past 15 years. In 2012, its analysis showed that 92 percent of awards had been collected by institutions tied to past and present directors. The figure dropped to 79 percent by this summer as the types of grantees have widened. Here is a sampling of conflict issues that have surfaced publicly over the years.
In 2007, violations involving five board members resulted in voiding applications from 10 researchers seeking $31 million. The applications included letters of support signed by deans of medical schools who also sat on the CIRM board of directors. Directors are barred from attempting to influence a decision regarding a grant. The agency blamed its employees for the problem.
In 2008, public complaints by one applicant from industry about conflicts of interest on the part of a reviewer were briefly aired at a public board meeting. The then chair of the CIRM board, Robert Klein, told the applicant the board needed instead to discuss naming CIRM-funded labs and then go to lunch. CIRM later refused to release the letter from the applicant detailing the problem.
In 2009, board member John Reed, then CEO of the Sanford-Burnham Institute, was warned by the state’s Fair Political Practices Commission about his violation of conflict of interest rules. Reed intervened with CIRM staff on behalf of a $638,000 grant to his organization. Reed took his action at the suggestion of then CIRM Chair Klein, an attorney who led the drafting of Proposition 71.
Also in 2009, then board member Ted Love, who had deep connections in the biomedical industry, served double duty for the agency. He was the interim chief scientific officer and helped to develop the agency’s first, signature $225 million disease team round while he was still serving on the board. As chief scientific officer, Love would have had access to proprietary information and trade secrets in grant applications.
When questioned, CIRM said that Love would serve only as a part-time advisor to the agency president, not as chief scientific officer. Nonetheless, in 2012, the board adopted a resolution with high praise for Love and his performance specifically as the chief scientific officer.
Beginning in 2010, a stem cell firm, iPierian,Inc., whose major investors contributed nearly $6 million to the ballot measure that created the stem cell agency, received $3.9 million in awards from the agency. The contributions were 25 percent of the total in the campaign, which was headed by Bob Klein. (See here and see here.)
In 2011, the chairman of the CIRM grant review group resigned from his position as the result of another violation, which the agency felt necessary to report to the California legislature. John Sladek, former president of Cal Lutheran University in Los Angeles, co-authored scientific publications with a researcher who was listed as a consultant on a CIRM grant application.
In 2012, StemCells, Inc., was awarded $40 million by the CIRM board despite having one of its $20 million applications rejected twice by grant reviewers. The action came after the board was vigorously lobbied by Klein, who had left his post as chair the previous year. Klein, who ran the Proposition 71 campaign, had campaign connections to researcher Irv Weissman of Stanford, who founded StemCells, Inc., and was on its board. Weissman was featured in a TV campaign ad for Proposition 71 and helped to raise millions for the 2004 ballot campaign.
The StemCells, Inc., awards were the first time that CIRM had approved that much money for one company, and the first time Klein lobbied his former board.
In 2012, an incident surfaced that illustrated how non-profit, disease-oriented organizations sometimes expect increased funding as the result of the appointment of sympathetic individuals to the board. That occurred when Diane Winokur was appointed to the board as a patient advocate. The chief scientist for The ALS Association, said Winokur will be “a tremendous asset in moving the ALS research field forward through CIRM funding.”
The IOM study identified as a problem the personal conflicts of interest involving the 10 patient advocates on the board. It said, “(P)ersonal conflicts of interest arising from one’s own or a family member’s affliction with a particular disease or advocacy on behalf of a particular disease also can create bias for board members.”
In 2013, internationally renowned scientist Lee Hood, winner of a National Medal of Science, violated the conflict of interest rules of the California stem cell agency when he was involved in reviewing applications in a $40 million round to create genomics centers in California. The conflict involved connections between Hood, Weissman and Trounson. It was not discovered by the agency during the closed-door review and was raised by another reviewer at the end of the review. The review had to be redone later in the year.
Hood never commented publicly, but CIRM said he acknowledged the conflict.
In January 2014, the genomics round surfaced again. The applications were by then before the CIRM board for public ratification of reviewers’ decisions. The reviewers’ actions are taken behind closed doors with no public disclosure of reviewers’ personal, professional or economic conflicts.
The genomics round riled some researchers who complained publicly in letters to the agency’s board about unfairness, apparent preferential treatment and manipulation of scores.
Only seven of the 29 members of the 29-member board could vote on the applications. Conflicts of interest and CIRM rules barred the rest from voting. The final vote on the award was 6-1 for a group led by Stanford. Two years earlier, however, when the “concept” plan was approved by the CIRM board, no directors were disqualified, even though some of their institutions were likely to benefit. The plan was approved on a show of hands. The transcript of the meeting does not indicate any negative votes or absentions.
The hidden review process
Under CIRM’s rules, the scientists who review the applications must come from out-of-state. They do not have to disclose publicly their economic, personal or professional conflicts despite the fact that they make the de facto decisions on the applications. The board rubber stamps nearly all of the reviewers’ actions to approve funding. A CIRM examination of the practice in 2013 showed that 98 percent of reviewers’ decisions were ratified by the board. Since then, the agency has not produced a similar report. Occasionally, however, the board will approve an application that was not recommended for funding.
The CIRM governing board has resisted requiring public disclosure of the interests of reviewers. The subject has come up several times, but board members have been concerned about losing reviewers who would not be pleased about disclosing their financial and other interests.
Nonetheless, public disclosure of economic interests among researchers is routine in scientific research articles. Many universities, including Stanford, also require public disclosure of financial interests of their researchers.
At the time of Hood-Weissman-Trounson flap, Stanford’s policy said, “No matter what the circumstances — if an independent observer might reasonably question whether the individual’s professional actions or decisions are determined by considerations of personal financial gain, the relationship should be disclosed to the public during presentations, in publications, teaching or other public venues.”
Proposition 71 placed the legal authority for grant approvals in the hands of the CIRM board. Traditionally in the world of science, other scientists (“ peer reviewers”), however, are deemed to be the most capable of making the scientific decisions about grant applications. The traditional practice calls for the reviewers to be anonymous and meet in private, which is also CIRM’s practice.
If the CIRM board concedes the decisions to the grant reviewers, state law is likely to require public disclosure of their financial interests, a move that the board has opposed for years. Former CIRM Chairman Klein repeatedly advised the board during its public grant approval processes that reviewers’ actions were only ”recommendations,” and that the board was actually making the decisions.
Proposition 14 implicitly recognizes, however, that a problem exists with directors approving “concept” plans for awards that could benefit their institutions.
To ease that problem legally, Klein inserted language in the new proposition that excludes adoption of “strategic plans, concept plans and research budgets” from being considered as matters involving conflicts of interest.
The measure does nothing to deal with matters involving the de facto, closed-door approval of awards by researchers who are unknown to the public and who do not have to publicly disclose their interests.
At the time the IOM report was released nearly eight years ago, some board members complained that its recommendations were unrealistic because of the likely, lengthy difficulties of altering a state law that had been created by the initiative. But since then, directors have not asked state lawmakers to change the structure of the board or to comply with the other $700,000 worth of IOM recommendations.
CIRM directors, however, missed an opportunity last year to seek conflict-easing changes through the $5.5 billion stem cell measure now on the ballot, Proposition 14.
Some board members have said they discussed the initiative privately with Bob Klein, who crafted the proposal last year.
Revision of CIRM’s conflict rules was discussed at a board meeting in May 2019. Several board members expressed concerns about the loss of valuable insights from board members who cannot vote on applications. Some also expressed concerns about whether loosening the rules would damage the possibility of voter approval of a ballot measure to refinance the agency. Several, including CIRM Chair Thomas, also said “there’s never been a conflict” involving a funding award and a board member. No action involving conflicts was taken at the meeting.
—
Editor’s Note: David Jensen is a retired newsman who has followed the affairs of the $3 billion California stem cell agency since 2005 via his blog, the California Stem Cell Report. He has published thousands of items on California stem cell matters in the past 11 years. This story was an excerpt from his upcoming book, California’s Great Stem Cell Experiment: Inside a $3 Billion Search for Stem Cell Cures, which s available for pre-order on Amazon.
Want to see more stories like this? Sign up for The Roundup, the free daily newsletter about California politics from the editors of Capitol Weekly. Stay up to date on the news you need to know.
Sign up below, then look for a confirmation email in your inbox.

[…] The conflicts of interests at the agency are posed by the nature of its 35-member governing board. It includes many persons with ties to institutions that receive awards. The situation was created by language in the two-ballot initiatives that created and funded CIRM. […]